Thin-Layer Chromatography: Comparative Estimation of Soil’s Atrazine
Vijay Kumar2 * , Niraj Upadhyay2 , Simranjeet Singh1 , Joginder Singh1 and Parvinder Kaur1
1
Department of Chemistry,
Lovely Professional University,
Punjab,
India
DOI: http://dx.doi.org/10.12944/CWE.8.3.17
Herbicide atrazine is a broad spectrum herbicide, used worldwide to protect the crops from weeds, but overuse of atrazine have caused the huge environmental problems from few decades. So it is very essential to study and develop the lab based analytical methods, which are important for the detection of atrazine in environment as well as in biological media. In this study we have collected the soils samples from farm fields and extracted the atrazine by Soxhlet method. Also we have extracted the atrazine from formulated grade considered as standard/pure sample in our study. Both extracted/standard and atrazine samples were characterized by UV and FTIR analysis. Further thin layer chromatography was run to check the purity of soil extracted sample.
Copy the following to cite this article:
Kumar V, Upadhyay N, Singh S, Singh J, Kaur P. Thin-Layer Chromatography: Comparative Estimation of Soil’s Atrazine. Curr World Environ 2013;8(3) DOI:http://dx.doi.org/10.12944/CWE.8.3.17
Copy the following to cite this URL:
Kumar V, Upadhyay N, Singh S, Singh J, Kaur P. Thin-Layer Chromatography: Comparative Estimation of Soil’s Atrazine. Curr World Environ 2013;8(3). Available from: http://www.cwejournal.org/?p=5129
Download article (pdf)
Citation Manager
Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2013-09-17 |
|---|---|
| Accepted: | 2013-10-23 |
Atrazine (2-chloro-4-ethylamino-6-isopropylamino-striazine), is probably the most commonly used chlorinated herbicide in the world.1 Atrazine is a selective systemic and most popular herbicide introduced in 1958 by J.R. Geigy. It has a range of trade names including Marksman, Coyote, Atrazina, Atrazol and Vectal. Atrazine is used for the pre and post-emergence control of annual and broad leaved weeds and perennial grasses; it inhibits photosynthesis and interferes with other enzymic processes.1,2 It is mainly absorbed through the plant roots, but can enter through the foliage, and accumulates in the apical meristems and leaves. Globally, atrazine is used in the production of maize, sorghum, sugar cane, pineapples, chemical fallows, grassland, macadamia nuts, conifers, and for industrial weed control, with its biggest market in maize production.2 The structure and physicochemical properties of atrazine are mentioned in table 1.1,2
As per literature atrazinehaving quite persistent in environment and is toxic to various living organisms.2 Number of methods have been described for the determination of atrazine in environment and biological media including, water and soils.3-7 Here we have reported the TLC based study, in this study, we have collected the atrazine contained soils samples from farm fields and extracted the atrazine from these soils samples by Soxhlet method. Also we have extracted the atrazine from formulated grade considered as standard/pure sample in our study. Both extracted/ standard sample and atrazine samples were characterized by UV and FTIR spectroscopy. Further thin layer chromatography was run to check the purity of soil extracted sample.
Experimental
TLC plates were prepared by pasting silica slurry on glass plates and dried at 1500C for 3 hours. TLC study was done in solvent system methanol–water (80:20). The soil samples were collected in zip bags from Phagwara City located in the North region of the India and Formulated atrazine, Tagtaf – 50 (Atrazine 50% WP) packets was purchase from local market. This sample area is representative of the agricultural region, mainly wheat, maize and paddy crop. The samples were collected at depths of 0-10, 10-30 and 30-50 cm from ten different points in an area of one hectare, and mixed to compose a single sample, for each depth. The sample was air-dried for three days, gently ground to pass through a 2 mm sieve and stored in a desiccator to be employed in the experiments. Soxhlet apparatus was set for the extraction of atrazine. Extraction time was 6 hrs at a rate of 4 cycles per hour for 5.0 g of the soil sample mixed with 2 g of Na2SO4, 200 ml of acetone was used for the extraction. The extract was evaporated to 10-15 ml by using a rotary evaporator {fig 1(b)}. Pure atrazine was extracted from formulated grade of atrazine by the liquid/solid phase extraction, using acetone. Further extracted atrazine passed through column to get the pure form, finally this obtained atrazine has been recrystalized using acetone/water in combination. This purified form characterized by UV and FTIR study and used as standard sample.
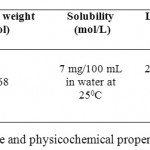 |
Table 1: Structure and physicochemical properties of Atrazine Click here to View table |
Results and Discussions
Randomly 10 soils samples were taken under study from the soil samples were obtained from twenty different collection points at depths ranging from 0 to 20 cm, and mixed thoroughly. Before use, the soils were air-dried and sieved through a 2.0 mm screen. The physical characteristics of the soils are presented in Table 2.
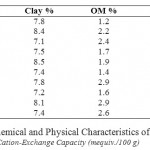 |
Table 2: Chemical and Physical Characteristics of the Soils. Click here to View table |
In the FTIR data (extracted from formulated grade currently used as pure sample after purification) the main peaks were observed in the region of 3200-3400cm-1 due to of secondary N-H stretching frequency and C=N and heterocyclic ring near 1450-1650cm-1 due to stretching frequency.8-10 In UV analysis ÊŽmax were observed at 225nm and 260nm π-π* and n-π* bands as in figure 1, because of ring transitions.9-11
 |
Figure 1: Comparative TLC spots of sample and standard of atrazine (a), Soxhlet setup for the etraction of atrazine from soils (b) and UV-visible spectra (in methanol) of extracted atrazine from formulated form of atrazine (c). Click here to View figure |
After running 10 TLC of extracted atrazine samples from soil {fig1(a)}, Rf values have been calculated and 2 ways ANNOVA was applied (using Origin 7.1 software). The following statistical data have been observed, as tabulated below. A linear results regression analysis of the values (samples) indicated an almost non linear fit of the data (r = 0.50). Therefore, t = 4.95134, p = 1.03165E-4 and at the 0.05 level, the two means are significantly different and results are significant.12-15 For the comparative observed values of samples and standard were complies approximate 75 i.e. F Statistic = 74.75 with slope, but -3.123 through origin.
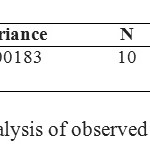 |
Table 3: Statistical analysis of observed TLC plates Rf values Click here to View table |
After setting a linear fit setting on samples of atrazine the observed equation was, Y = 0.480X + 0.472 and R2 = 0.5043. It has been observed that there was high level of variations among the samples as compared to standards.13-17
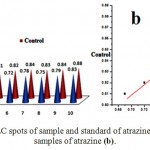 |
Figure 2: Comparative TLC spots of sample and standard of atrazine (a), and linear fit plot for samples of atrazine (b). Click here to View figure |
Conclusion
On the basis of these results, it is difficult to equate pesticide applications on soil thin-layer chromatography with field rates. It can be seen that the methodology applied to classify the mobility of the herbicide atrazine was efficient and showed advantages in simple lab based methods to predict quantitatively the leaching and amount of decomposition of herbicides. A linear results regression analysis of the values (samples) indicated an almost linear fit of the data (r = 0.50) with low precision.
References
- TB Hayes, A Collins, M Lee, M Mendoza and A Vonk, PANS, 99, 5476 (2002).
- Environmental Protection Agency (EPA), Interim Reregistration Eligibility Decision for Atrazine, U.S. Environmental Protection Agency, Washington, DC, 2003.
- S Navarro, N Vela, C Garcıa and G Navarro, J. Agric. Food Chem, 51, 7359 (2004).
- Z. Vryzas and E. Papadopoulou-Mourkidou, J. Agric. Food Chem, 50, 5026, (2002).
- E. Turiel, A Martin-Esteban and P Fernandez, Anal. Chem, 73, 5133 (2001).
- S. Stipicevic, S. Fingler, L Zupancic-Kralj and V Drevenkar, J. Sep. Sci, 26, 1237 (2003).
- MEC Queiroza and FM Lançasb, J. Env. Sci Health, Part B, 35, 467 (2000).
- http://webbook.nist.gov/cgi/cbook.cgi?ID=C1912249&Mask=80.
- Donald L. Pavia, Gary M. Lampman, George S. Kriz, and James R. Vyvyan, Introduction to Spectroscopy, 4th Ed. 2009 Brooks/Cole, Cengage Learning, Nelson Education Ltd Canada.
- Silberstein RM, Webster FX and Kiemle DJ, 2005 Spectroscopic Identification of Organic Compounds, 7th Ed. 2005, Johan Willy & Sons Inc.
- M. Cea, P. Cartes, G. Palma, M.L. Mora, R.C. Suelo Nutr. Veg, 10, 62 (2010).
- E. Gonza´lez-Pradas, M. Villafranca-Sanchez, F. Del Rey-Bueno, M.D. Uren˜a-Amate, M. Ferna´ndez-Pe´rez, Pest Manag Sci, 56, 565 (2000).
- M. H. Guermouche, D. Habel, S. Guermouche, J.AOAC Int. 82, 244 (1999)
- R. J. Vanhaelen-Fastré, M. L. Faez, M. H Vanhaelen, J. Chromatogr, 868, 269 (2000.
- B. Simonovska, S. Andrensek, I. Vovk, M. Prosek, J. Chromatogr, 862, 209 (1999).
- R. K. Sarin, G. P. Sharma, K. M. Varshney, S. N. Rasool, J. Chromatogr, 822, 332 (1998).
- J. Stuart Hunter, J. Assoc. Off. Anal. Chem, 64, 574 (1981).






