A Comparative Study of Removal of Paraphenylene Diamine Using Bentonite and Activated Charcoal
1
Department of Chemistry,
Tilka Manjhi Bhagalpur University,
Bhagalpur,
Bihar
India
2
Atomic Minerals Directorate for Exploration and Research,
Bangalore,,
Karnataka
India
Corresponding author Email: ashokjha39@gmail.com
DOI: http://dx.doi.org/10.12944/CWE.20.2.12
Copy the following to cite this article:
Anand A, Jha A. K, Thakur R. A Comparative Study of Removal of Paraphenylene Diamine Using Bentonite and Activated Charcoal. Curr World Environ 2025;20(2). DOI:http://dx.doi.org/10.12944/CWE.20.2.12
Copy the following to cite this URL:
Anand A, Jha A. K, Thakur R. A Comparative Study of Removal of Paraphenylene Diamine Using Bentonite and Activated Charcoal. Curr World Environ 2025;20(2).
Download article (pdf)
Citation Manager
Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2025-06-26 |
|---|---|
| Accepted: | 2025-09-01 |
| Reviewed by: | 
 Parwathi Pillai
Parwathi Pillai
|
| Second Review by: |

 Valentí Turu Michels
Valentí Turu Michels
|
| Final Approval by: | Dr. Mohammad Rafatullah |
Introduction
Water is one of the basic elements for the formation of living organisms, so studying water and its investments is extremely important for the better sustainability of life in all aspects. Water pollution is defined as a change that occurs in the natural characteristics of water, directly or indirectly, due to human activity, which makes it unfit for various uses as confirmed by the World Health Organization. Water is polluted by human, animal, plant, mineral, Industrial or chemical waste, heavy metals or toxic dyes such as para-phenylenediamine (PPD).1
P-Phenylenediamine occurs as a white to slightly red solid or crystal that darken on exposure to air. It is slightly soluble in water. Paraphenylenediamine (PPD), a component of hair dyes is a p-nitro aniline derivative.2-3 Apoptosis is induced through reactive oxygen species generated by PPD. The proposed mechanism of carcinogensis induced by the dye is through production of reactive oxygen species, oxidative stress and binding with DNA. In addition PPD induces allergic dermatitis. Skin irritation and renal impairment may also take place by PPD. Oxidative stress may also be introduced in different cells by PPD.
It is commonly, used in the dyeing industry, particularly in hair coloring products. PPD poses potential health risks to humans, cause respiratory problems, asthma, allergic reactions, leading to skin irritation, rashes and potential carcinogenic effects during dealing, using or exposure.4-5 PPD has also got its use in rubber and polymer industries. For dyeing of hair, PPD is mixed with H2O2 to get the oxidation product which help formation of permanent bond after reaction with sulfihydryl groups in the hair. P-phenylene diamine gives black color on oxidation.
Degradation
Natural dyes are preferred to synthetic dyes due to harmful toxic effects. Anionic, cationic and non-ionic dyes are also used in dyeing industries. Oxidation and hydrolysis are the processes of degradation of dyes. Agriculture byproducts, wastes, and activated charcoal prepared from biomass have been used for degradation of organic dyes from waste water effluents from textile industries.6-8
Electrocoagulation has also been used as one of the methods of removal of paraphenylene diamine.9 Activated charcoal has been modified for effective degradation of few organic dyes.10 Green biosorbents and few clay minerals have also been established as an effective adsorbent of organic dyes in general and paraphenylene diamine particular.11-12 Bentonite, powder has been utilized in the present paper for degradation of PPD due to exchangeable cations and high surface area.13-15 Bentonite minerals have industrial applications such as decolorisation of oil, wine, capping of holes in oil wells and pharma companies.16 Rajasthan as well as Rajmahal bentonite have the potential to remove, Cr(VI), Pb(II), As, and malachite green from the aqueous medium.17 Bentonite mineral of Rajmahal hill has got specific industrial applications due to the presence of traces of rare earth metals and high surface area.18-20 Percentage removal of paraphenylene diamine has been recorded using bentonite and active charcoal in the present paper.
Materials and Methods
The stock solution of 100 ppm para-phenylenediamine(PPD) is prepared by dissolving PPD in hot water and then the 80ppm,50ppm and 20ppm solution is prepared by dilution method V1N1= V2 N2 . The pH is adjusted from 7 to 4 and by addition of N/10 HCL and N/10 NaOH recpectively. 100 ml of 100ppm PPD solution is treated with 1g bentonite powder of 300 mesh sieve at a regular interval of 15 minutes. The residual concentrations are known by UV double beam spectrophotometer Systronics AU 2703. The magnetic stirrer was used in the experiment for stirring at an rpm of 220. Rajasthan bentonite (Barmer) has been used as an adsorbent of dye. The bentonite has been powdered to 300 mesh sieve dried at 60º in an oven and used for decoloration of the dye. The degdration of the dye was studied at different parameters and the absorbance of the degraded sample was noted at hmax = 307 nm. The same experiment was repeated with different concentrations of PPD at different PH values such as 4 and 9. One gram bentonite powder was treated with the PPD solution up to a fixed time interval of 15 minutes and the residual concentrations of PPD were recorded.
PPD was procured from Merck company and deionized water has been used for preparing the solution of PPD of different concentrations. The cation exchange capacity of bentonite has been determined by Na2CO3 method. The cation exchange capacity has been determined to be 90 miliequivalent/litre. The FTIR of bentonite samples has been done before and after adsorption. The chemical composition of bentonite has been found to contain major and minor oxides such as SiO2 , Al2O3,K2O,MgO,TiO2.
Results
In case of para phenylenediamine(PPD), it was observed that the dye was absorbed on the surface of bentonite minerals .
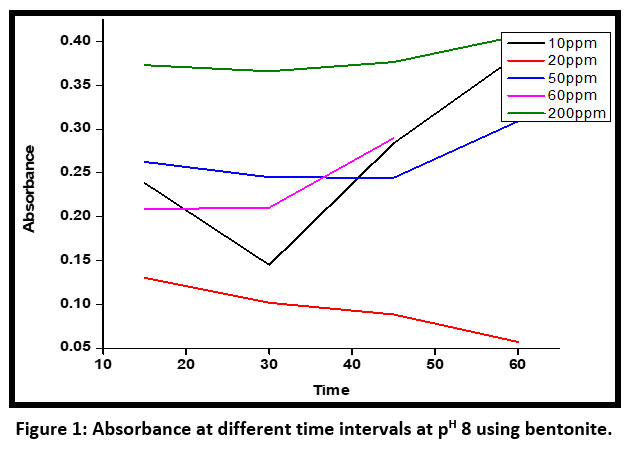 | Figure 1: Absorbance at different time intervals at pH 8 using bentonite.
|
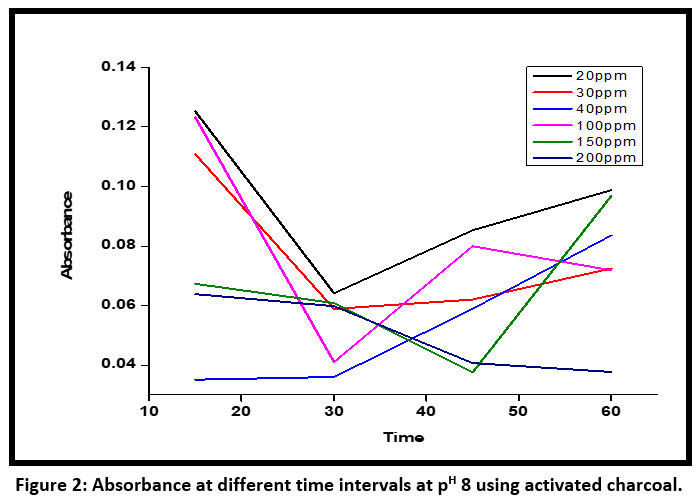 | Figure 2: Absorbance at different time intervals at pH 8 using activated charcoal.
|
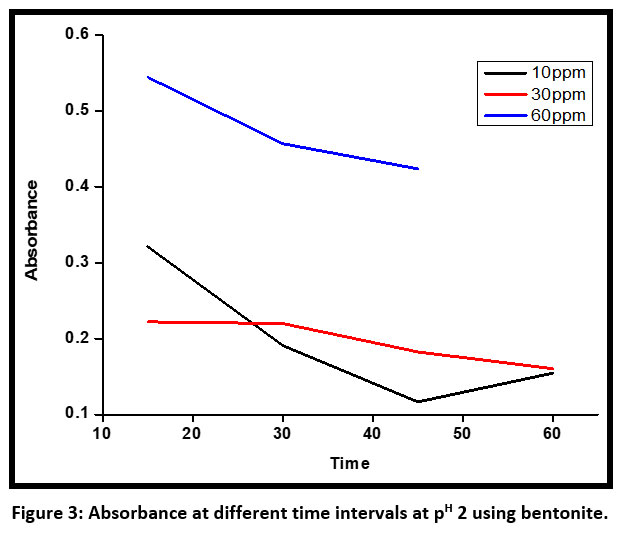 | Figure 3: Absorbance at different time intervals at pH 2 using bentonite.
|
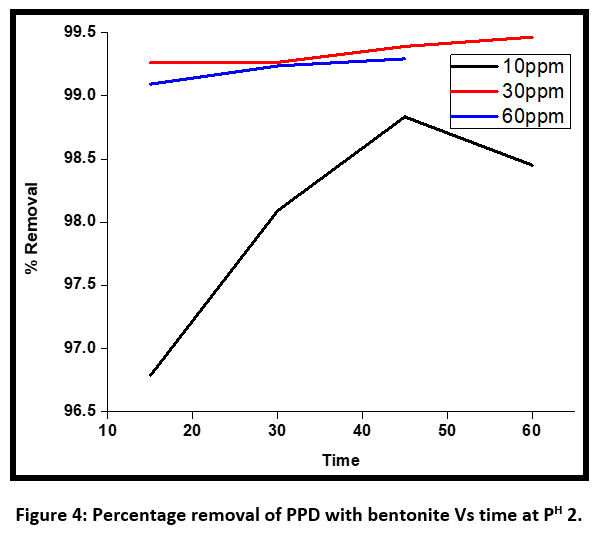 | Figure 4: Percentage removal of PPD with bentonite Vs time at PH 2 .
|
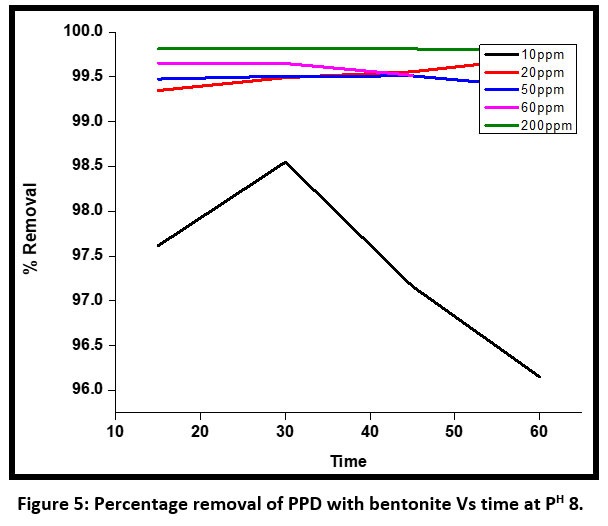 | Figure 5: Percentage removal of PPD with bentonite Vs time at PH 8 .
|
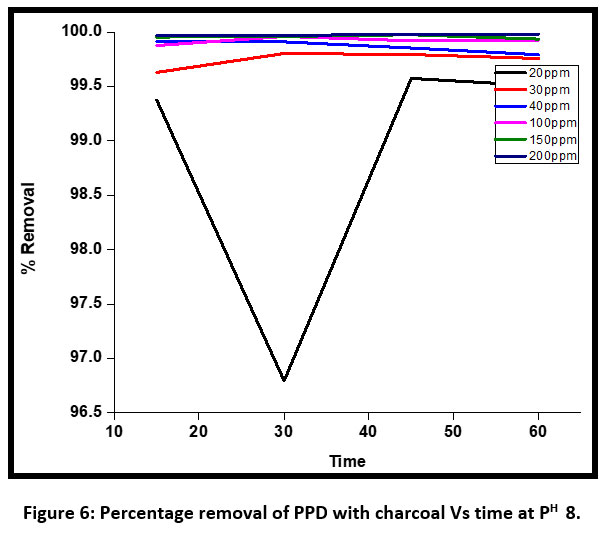 | Figure 6: Percentage removal of PPD with charcoal Vs time at PH 8 .
|
The percentage dye decolorization was calculated using the formula.
![]()
Decolorisation % = ×100
Ao = Initial absorbance
At = Absorbance at time t
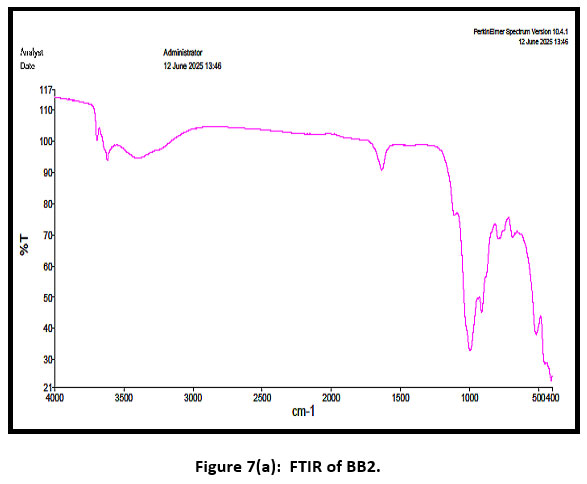 | Figure 7(a): FTIR of BB2 .
|
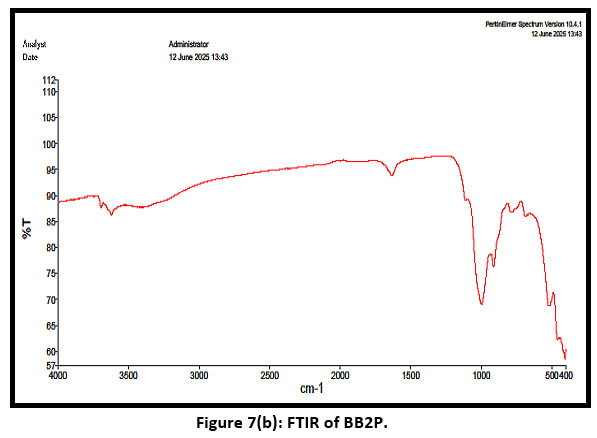 | Figure 7(b): FTIR of BB2P.
|
In figure 7(a), it shows the fourier transform infrared (FTIR) fix of Bentonite. The peak around 3600 cm-1 confirms the presence of -OH whereas the peak around 1630 cm-1 confirms Si-O-Si stretching vibration. The Al–O bond may be attributed to the peak at 915 cm-1.
In figure 7(b), it shows the peaks after adsorption. The shift in peaks and intensity clearly confirmed the adsorption of the dye. Strong peaks at 1060 cm-1 and 540 cm-1 confirm the presence of montmorillonite unit in bentonite so, the adsorption is due basically by the montmorillonite presence. .
Percent decolorisation was plotted against time at a regular interval of 10 minutes up to 60 minutes. Time interval of 10 minutes, 20 minutes, 30 minutes, 40 minutes, 50 minutes and 60 minutes for an initial concentration of 10 ppm, 30 ppm and 60 ppm at PH 2. Similar experiments were repeated and percent discoloration plotted against same time intervals at pH 8 for concentrations of 10 ppm , 20 ppm, 50 ppm, 60 ppm and 200 ppm.
Discussion
The adsorbance of dye on the bentonite surface has been attributed to the high surface area of bentonites and cation exchange capacity also. At an alkaline pH maximum absorbance has been observed for an initial concentration of 10 ppm. For an initial concentration of 60 ppm, the absorbance decreases when the time increases from 10 minutes to 60 minutes using bentonite as an adsorbent(Figure – 1).
Absorbance using activated charcoal at PH 8, has been recorded for initial concentrations of 20 ppm ,30 ppm, 40 ppm ,100ppm , 150 ppm and 200 ppm respectively (Figure – 2).
Figure 3 shows absorbance at different time intervals at PH 2 using bentonite for initial concentrations of 10 ppm , 30 ppm and 60 ppm at time intervals of 10, 20, 30, 40, 50, and 60 minutes respectively. At an acidic pH of 2 and initial concentration of 10 ppm the absorbance of the dye decreases regularly but for an initial concentration of 60 ppm, the absorbance decreases and then increases abruptly at around 45 minutes. This clearly shows that some release of para-phenylene diamine(PPD) takes place.
A kinetic study was done to find out the optimum condition. The discoloration of the dye was much faster initially for an initial concentration of 10 ppm PPD at PH 2 and after 45 minutes of contact time the percentage removal decreased (Figure 4). At an alkaline pH 8 the percentage removal was up to 98 % at 30 minutes and then decreased up to 60% . So the optimum time at pH for discoloration of dye was observed at 30 minutes.
Figure 6 shows the percentage removal of PPD with active charcoal Vs time at pH 8. At this pH the discoloration percentage of dye is 100% complete for initial concentration of 30 ppm, 40 ppm, 100 ppm, 150 ppm, and 200 ppm respectively. Maximum removal of PPD has been observed at pH 8 using active charcoal.
A comparative study of decoloration of PPD by charcoal and bentonite showed that both of them had the discoloration potential but discoloration percentage using activated charcoal is maximum at pH 8 having different initial concentrations.
During the initial period the discoloration percentage increased but the percentage discoloration using active charcoal was almost 100% for all initial concentrations except 20 ppm. The equilibrium was achieved at 15 minutes and, the discoloration percentage remained the same starting from 20 minutes to 60 minutes at a regular interval of 10 minutes. An alkaline pH of 8 is the best fit for decoloration of PPD using active charcoal.
Figure 5, clearly stated that for an initial concentration of 300 ppm the discoloration percentage was maximum using bentonite as an adsorbent within a range of 10 minutes to 60 minutes. For an initial concentration of 10 ppm, the discoloration percentage first increased up to 30 minutes and then decreased after 30 minutes up to 60 minutes at PH 8 using bentonite powder. The bentonite is regenerated after washing the adsorbed material several times by hot water.
Conclusion
Bentonite minerals can be used as an excellent adsorbent of paraphenylene diamine (PPD). Bentonite minerals can be recycled for use by washing several times with deionized water because hot water dissolves the adsorbate and bentonite becomes free of adsorbate.
Acknowledgement
The authors acknowledge the facility of ICP-AES extended to us by Dr. Raghbendra Thakur, D.A.E, Banglore.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects or any material that requires ethical approval.
Informal Consent Statement
This study did not involve human participants, and therefore informal consent was not required.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Amit Anand: Conducted the analytical tests for absorbance of PPD.
Ashok Kumar Jha: Prepared the manuscript.
Raghbendra Thakur: DAE Banglore made available the facility of ICP-AES.
References
- Environmental Protection Agency. Health and Environmental Effects Profile for Phenylenediamines. Environmental Criteria and Assessment Office. Office of Health and Environmental Assessment. Office of Research and Development. Cincinnati,OH,USA.1985.
- Stanley, L. A., Skare, A.J., Doyle,E., Powrie,R., D’Angelo,D., Elcombe,C.R. Lack of evidences for metabolism of p-phenylenediamine by human hepatic cytochrome P450 enzymes. Toxicology. 2005;210(2-3): 147-157. DOI: 10.1016/j.tox.2005.01.019.
CrossRef - Huncharek, M., Kupelnick,B. Personal of hair dyes and the risk of bladder cancer: Results of a Meta-Analysis. Association of schools of public health; 2005. 120(1): 31-38. DOI : https://doi.org/10.1177/003335490512000107
CrossRef - Hilton, C.L. Spectrophotometric determination of flexzone 3C and other p-phenylene diamine derivatives. Analytical Chemistry. 1960; 32(12):1554-1557. DOI: 10.1021/ac60168a005.
CrossRef - Cui, H., Dong, S., Wang, K., Luan, M., Huang, T. Synthesis of a novel Type-II In2S3/Bi2MoO6 heterojunction photocatalyst: excellent photocatalytic performance and degradation mechanism for Rhodamine B. Separation and Purification Technology. 2021; 255: 117758. DOI: 10.1016/j.seppur.2020.117758.
CrossRef - Cazetta, A.L., Pezoti, O., Bedin, K.C., Silva, T.L., Junior, A.P. et al. Magnetic activated carbon derived from biomass waste by concurrent synthesis: efficient adsorbent for toxic dyes. ACS Sustainable Chemistry and Engineering. 2016; 4(3): 1058-1068. DOI: 10.1021/acssuschemeng.5b01141.
CrossRef - Gumus, D., F. Akbal. Photocatalytic degradation of textile dye and wastewater. Water, Air and Soil Pollution. 2010; 216: 117-124. DOI: 10.1007/s11270-010-0520-z
CrossRef - Nekouei, F., Shahram, N., Tyagi, I., Gupta, V. K. Kinetic, thermodynamic and isotherm studies for acid blue 129 removal from liquids using copper oxide nanoparticle-modified activated carbon as a novel adsorbent. Journal of Molecular Liquids. 2015; 201: 124-133. DOI: 10.1016/j.molliq.2014.09.027
CrossRef - Fuat, O., Gokkus, O., Sabuni,M., Removal of disperse and reactive dyes from aqueous solutions using ultrasound- assisted electrocoagulation. Chemosphere. 2020; 258: 127325. DOI: http://dx.doi.org/10.1016/j.chemosphere.2020.127325
CrossRef - Goswami, R., Dey, A.K. Use of anionic surfactant- modified activated carbon for efficient adsorptive removal of crystal violet dye. Adsorption Science and Technology. 2022; 2022(4): 1-28. DOI: https;//doi.org/10.1155/2022/2357242
CrossRef - Parlayici, S. Green biosorbents based on glutaraldehyde cross linked alginate/sepiolite hydrogel capsules for methylene blue, malachite green and methyl violet removal. Polymer Bulletin. 2023; 80(3): 2457-2483. DOI: 10.1007/s00289-022-04174-6
CrossRef - Shojaei, S., Nouri, A., Baharinikoo, L., Farahani, M.D., Shojaei, S. Removal of the hazardous dyes through adsorption over nanozeolite-X: simultaneous model, design and analysis of experiments. Polyhedron . 2021; 196: 114995. DOI: 10.1016/j.poly.2020.114995
CrossRef - Jha, A.K., B. Mishra. Removal of fluoride by bentonite minerals of Rajmahal Hills. Journal of Indian Chemical Society . 2012; 89(4): 519-521.
- Pandey, S., Ramontja, J. Natural bentonite clay and its composites for dye removal: Current state and future potential. American Journal of Chemistry and Applications. 2016; 3(2): 8-19.
CrossRef - Zdravkovic, A.S., Stankovic, N.J., Ristic,N.N., Petkovic,G.M. Application of activated bentonite for the removal of direct and reactive dye from aqueous solutions. Chemical Industry & Chemical Engineering Quarterly. 2019; 25(4): 341-351. DOI: 10.2298/CICEQ171025012Z
CrossRef - Jha, A.K. Bentonite for Chemical Industries. Journal of Indian Chemical Society. 2018; 95(1): 35-40.
- Majumder,S., Jha, A.K. Removal of Cr and Mn from aqueous medium using bentonites and their derivatives. Journal of Chemical Science. 2020; 132:135 . DOI: 10.1007/s12039-020-01838-6
CrossRef - Sikder,S., Jha,A.K., Thakur,R. Rare earth and trace metal characteristics of bentonite in the Rajmahal hills. ES Materials and Manufacturing. 2023;20(840):1-6. DOI: 10.30919/esmm5f840
CrossRef - El-Nagar, Doaa A., Halim, K.Y.A. Remediation of heavy metals in contaminated soil by using nano-bentonite, nano-hydroxyapatite, and nano-composite. Land Degradation & Development. 2021; 32(16): 4562-4573. DOI: 10.1002/ldr.4052
CrossRef - Romero,L.E.L., Gaona,M.S., Vargas,C.A.P., Perez,D.L., Amaya,J. Zero-valent iron nanoparticles synthesized by ultrasound for the adsorption of malachite green dye. Catalysis Today. 2025; 459:115417. DOI: 10.1016/j.cattod.2025.115417.
CrossRef






