Studies on Biodegradation of Acetaminophen by Bacillus subtilis subsp. subtilis NCIB 3610(T)
Meghmala Sheshrao Waghmode
*
 , Snehal Balu Lende
, Pranali Ratnakar Gaikwad
, Snehal Balu Lende
, Pranali Ratnakar Gaikwad
 , Neha Nitin Patil
and Ujwala Vinayak Khisti
, Neha Nitin Patil
and Ujwala Vinayak Khisti

1
Department of Microbiology,
PDEA’s Annasaheb Magar Mahavidyalaya,
Hadapsar, Pune,
Maharashtra
India
Corresponding author Email: meghmicro@gmail.com
DOI: http://dx.doi.org/10.12944/CWE.18.1.14
Copy the following to cite this article:
Waghmode M. S, Lende S. B, Gaikwad P. R, Patil N. N, Khisti U. V. Studies on Biodegradation of Acetaminophen by Bacillus subtilis subsp. subtilis NCIB 3610(T). Curr World Environ 2023;18(1). DOI:http://dx.doi.org/10.12944/CWE.18.1.14
Copy the following to cite this URL:
Waghmode M. S, Lende S. B, Gaikwad P. R, Patil N. N, Khisti U. V. Studies on Biodegradation of Acetaminophen by Bacillus subtilis subsp. subtilis NCIB 3610(T). Curr World Environ 2023;18(1).
Download article (pdf) Citation Manager Publish History
Select type of program for download
| Endnote EndNote format (Mac & Win) | |
| Reference Manager Ris format (Win only) | |
| Procite Ris format (Win only) | |
| Medlars Format | |
| RefWorks Format RefWorks format (Mac & Win) | |
| BibTex Format BibTex format (Mac & Win) |
Article Publishing History
| Received: | 2022-10-03 |
|---|---|
| Accepted: | 2023-02-20 |
| Reviewed by: | 
 Illyas bin Md Isa
Illyas bin Md Isa
|
| Second Review by: |

 Essam S. Soliman
Essam S. Soliman
|
| Final Approval by: | Dr. Mohammad Oves |
Introduction
The presence of pharmaceutical contaminants in the aquatic environment has become a subject of emerging concern1. The pharmaceuticals contaminants comprising drugs, heavy metals, dyes, and personal care products have been considered emerging environmental contaminants due to their occurrence in water bodies2,3. Now, many of these compounds are perceived worldwide ranging from ng/L to ?g/L but due to their biologically active potential, it could affect the ecosystem. With the increased production and consumption, micropollutants and personal care products are occurring in the environment.
One of the most frequently detected pharmaceutical painkillers in treated and untreated waste water is acetaminophen (APAP; paracetamol; N-acetyl-para-aminophenol)4. Acetaminophen is a heavily demanded over the counter medicine. Paracetamol is prescribed as fever reductant, pain reliever, antipyretic, and analgesic drug5. Acetaminophen is continuously entered into the aquatic environment by many emission routes starting from industrial wastewater, customer use and clearance and hospital discharged water 6,7, 8. Wastewater treatment plants aim to remove of toxicants leading to the production of intermediates or end products that are highly ecotoxic related to the parent molecule.
The COVID-19 pandemic is predicted to have a big effect on the use of acetaminophen and subsequently its occurrence in the environment. At the wastewater treatment plants, advanced methods like electrochemical oxidation9, H2O2/UV; H2O2/Fe2+/UV oxidation10 ,ozonation11, and photo-catalysis methods12 are used to remove paracetamol like drugs . Physico-chemical methods have high operation costs and give the possibility of the generation of toxic intermediate or end products. Biological treatment can be employed for the removal of pharma micropollutants to reduce the risk factors associated with other methods. For the biological methods, the selection of microorganisms based on their degradative potential, biodegradative pathway and cultivation conditions, is the first step that must be studied. This studies was aimed to isolate Bacillus subtilis subsp.subtilis and using it for the biodegradation of acetaminophen.
Materials and Methods
Chemicals used and collection of samples
The standard of Paracetamol was purchased from Loba Co. Ltd. The pharmaceutical wastewater was used for the isolation of acetaminophen degraders. This sample contains untreated industrial waste water. The five samples were kept in the sterile containers during transportation to the laboratory and then samples were stored at 4 °C. The samples were kept for the settling of heavy particles like sand, and other debris. The sediments were discarded before use and the upper layer of water was used for the further experiment.
Enrichment, screening, isolation, and identification of APAP?degrading Bacteria
APAP degradative strains isolation was carried out as per the protocol of Chopra and Kumar, with little modification13. APAP biodegradation test was performed in 250-mL Bushnell Haas Medium (BHM) containing acetaminophen and waste water sample and was incubated at room temperature for 3 days. The microorganisms were isolated by culturing on a nutrient agar plate and Bushnell Haas medium with acetaminophen (100 -3000ppm). The isolate showing maximum tolerance to acetaminophen, was used for further studies. The isolated bacteria were classified using the phenotypic and genotypic characteristics. The characterizations have been grounded on morphological, biochemical tests and 16s rRNA sequencing method for the confirmation.
Fermentation Studies
The isolate was inoculated in Bushnell Haas broth (PH 7) containing APAP at a rate of 2500 ppm. The flasks were incubated at room temperature in dark conditions for 4 days with an intermittent determination of cell concentration (OD 600nm), PH and residual concentration of APAP using colorimetric method14. The standard dose response curve of paracetamol 100-1000 ug/mL was used as a reference for the calculation. The biodegradation of APAP was calculated by Eq. 1:
Rate of degradation (%) = (C0-Ct)/C0 × 100 (1)
Where C0 is the preliminary concentration of APAP; Ct is the concentration of APAP after incubation at time ‘t’. Kinetic parameters during biodegradation were evaluated using Computer Assisted Kinetic Evaluation (CAKE) program34.
Reverse-phase HPLC based analysis
The fermented broth was centrifuged and the supernatant was collected. Solvent extraction was carried out using methanol. After solvent evaporation,the sample was processed for reverse phase HPLC. The elution of paracetamol was carried out with the assistance of reverse phase HPLC by isocratic mode, JASCO (250 × 4.6 mm, packed with 5 µ). The solvent system used was methanol: water (15:85) with 1 ml/min flow rate and monitored at 240 nm.
Identification of APAP degradative metabolite
The analysis of APAP biodegradative metabolite was carried out using High-Resolution Mass Spectrometry (HR-MS). Bruker Impact HD instrument was used for the mass analysis of the standard and its biodegradative metabolite extracted from the fermented broth. HR MS was done with conditions of a double electrospray ionization source in positive ion mode , scanning range of 50 to 1200 m/ z , 4.0 spectra/sec of scan rate, 200°C source gas temperature, 7.01 l.min-1 gas flow rate and 10 ?l sample injection volume. The Nebulizer pressure was adjusted to 1.7 bars. Data were analyzed with the assistance of Bruker compass data analysis 4.2 Software.
Screening of Biodegradation of Acetaminophen Bioaugmentation by employing chemotaxis assay
The chemotactic behavior of the strain towards acetaminophen was tested qualitatively with the help of drop plate assay according to the procedure described earlier15. The medium was smeared with bacterial inoculum on the agar surface of medium. A pinch (5-10 mg) of acetaminophen crystals were placed in the well (6 mm) of the plate. The plate was incubated at room temperature for 48 Hrs. and bacterial growth near the chemical was recorded as a positive chemotactic response.
Results
Enrichment, screening, isolation, and identification of APAP?degrading Bacteria
Enrichment of acetaminophen degraders was done using Bushnell Haas Medium containing acetaminophen (100-3000mg/L) concentration. Bacterial Cultures showing maximum tolerance to acetaminophen were sub cultured on nutrient agar plates. A total of 23 isolates were obtained but isolates showing high APAP tolerance were further studied.
The isolate showing tolerance to 2500 ppm concentration of APAP was found to be Gram-positive rod-shaped bacteria with cream-colored colonies. Based on the biochemical tests, isolate found to be catalase positive, glucose fermentative, methyl red positive and non-spore forming. Molecular identification was done by 16S rDNA sequencing method. Isolate was found to be Bacillus subtilis subsp. subtilis NCIB 3610(T) with 99.86% similarity using EzBioCloud Database. Phylogenetic dendrogram (Fig.1) was plotted using the neighbor joining method.
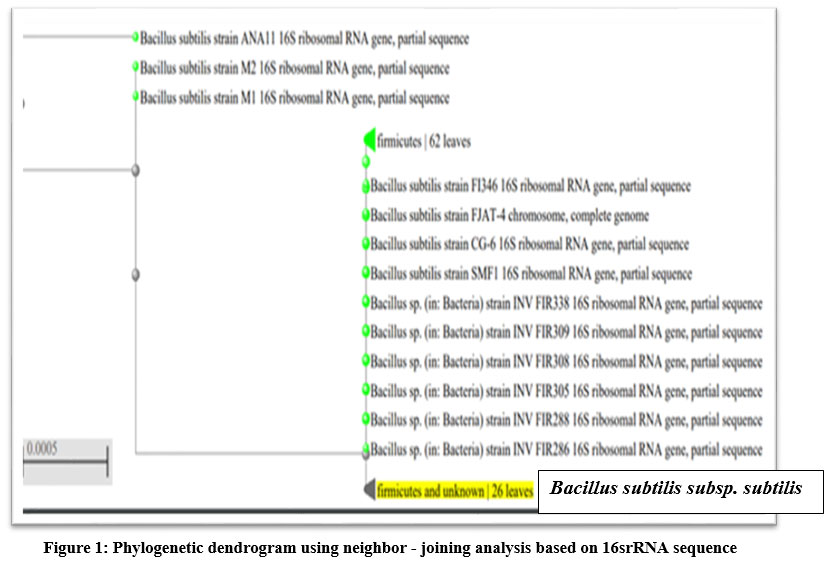 | Figure 1: Phylogenetic dendrogram using neighbor - joining analysis based on 16srRNA sequence.
|
Fermentation Studies
The isolate was inoculated in BHM medium (pH 7) containing acetaminophen (2500ppm) concentration and1% inoculum (109 cfu/ml).Flasks were incubated at room temperature for 04 days to evaluate the time course effect with the relationship between acetaminophen reduction associated with bacterial growth. With the increase in the incubation period, the broth showed brown to black coloration as shown in Fig.2.
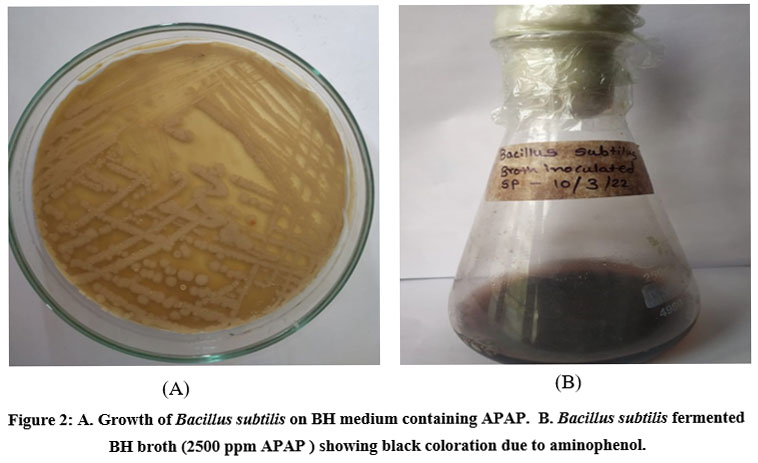 | Figure 2: A. Growth of Bacillus subtilis on BH medium containing APAP. B. Bacillus subtilis fermented BH broth (2500 ppm APAP ) showing black coloration due to aminophenol.
|
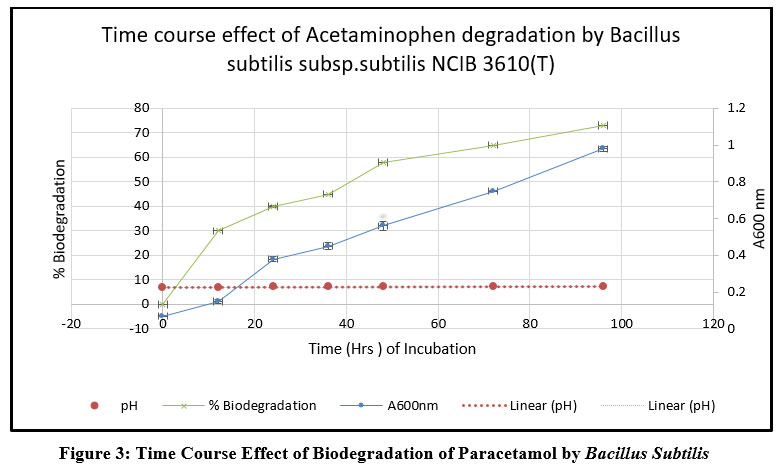 | Figure 3: Time Course Effect of Biodegradation of Paracetamol by Bacillus Subtilis.
|
The strain Bacillus subtilis degraded 73 % of acetaminophen with an increase in biomass and Ph as given in Fig.3. The specific growth rate of the Bacillus subtilis was calculated using Monod model as 0.3499, when grown in the presence of acetaminophen. Kinetic parameters during biodegradation was evaluated using Computer Assisted Kinetic Evaluation (CAKE) program and results are given in Table.1.
Table 1: Kinetic parameters for APAP biodegradation by Bacillus subtilis
Model Kinetic | APAP concentration | K (d-1) | Chi-sq error | DT50 (days) | DT90 (days) |
Simple First Order | 2500 ppm | 0.1 | 5.1 | 3.4 | 11.5 |
Reverse-phase HPLC- based analysis
Reverse-phase HPLC was done with the standard paracetamol and biotransformed metabolite of paracetamol extracted from Bacillus Subtilis sp. fermented broth given in Fig.4a and 4b. In the HPLC chromatogram of the biotransformed metabolite of paracetamol, alongwith the paracetamol (6.4 min retention time), one unknown metabolite was found with a retention time of 2.217 min. Bacillus subtilis degraded 73.4% acetaminophen (2500ppm) after 96 hours of incubation.
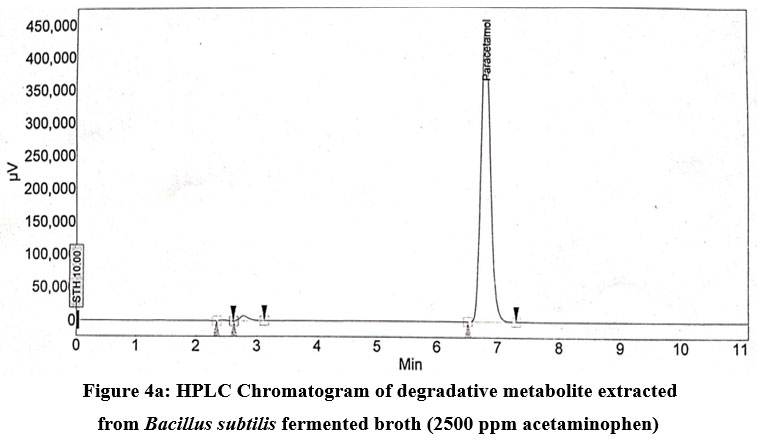 | Figure 4a: HPLC Chromatogram of degradative metabolite extracted from Bacillus subtilis fermented broth (2500 ppm acetaminophen).
|
 | Figure 4b: HPLC chromatogram of standard acetaminophen (Paracetamol).
|
Identification of APAP degradative metabolite
Methanol -extracted degradative metabolite of acetaminophen was identified using high -resolution mass spectroscopy. One peak of 116.10 of 4 aminophenol (C6H7NO) was identified as the biodegradative product of acetaminophen using Bruker compass data analysis 4.2 Software (Fig.5).
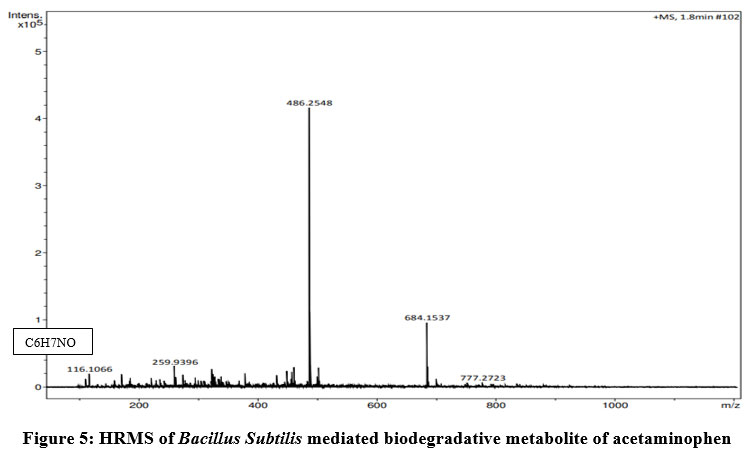 | Figure 5: HRMS of Bacillus Subtilis mediated biodegradative metabolite of acetaminophen.
|
Screening of Biodegradation of Acetaminophen Bioaugmentation using chemotaxis assay
The positive - chemotactic behavior of Bacillus subtilis towards paracetamol was studied by using drop plate assay (Fig. 6). Drop plate assay result showed the development of bacterial ring near the crystals of acetaminophen after 48 Hrs. of incubation suggesting positive chemotactic behavior of the strain which is the primary requirement for bioaugmentation.
 | Figure 6: Chemotaxis of Bacillus Subtilis towards acetaminophen .
|
Discussion
Bacillus subtilis is a Gram-positive bacterium that is well known for its potential application in bioremediation. The biodegradation of pharmaceutical micropollutants like paracetamol has gained interest among researchers due to low cost and efficacy. Bacteria viz., Rhodococcus ruber, Stenotrophomonas, Pseudomonas moorei, Delftia tsuruhatensis, Cunninghamella echinulate, and Shinella has been reported to have paracetamol biodegradative potential 16, 17 ,18,19. In Bacillus genera, Bacillus drentensis and Bacillus cereus have been reported to degrade acetaminophen at a rate of 300 and 200 ppm respectively 13,21. Compared to this reported strain, Bacillus subtilis subsp.subtilis NCIB 3610(T) was found to degrade paracetamol in a higher concentration of 2500 ppm ,giving a more ecotoxic end product 4 Aminophenol. Sewage wastewater isolate Bacillus subtilis strain DPP3 (MN744327) followed Haldanes growth kinetic model with the maximum specific growth rate of 0.259 (Hr -1) in the presence of 593 ppm concentration of acetaminophen and 10 days incubation period for the degradation33. The paracetamol biodegradation pathway comprises the catalytic activity of hydroquinone 1,2-dioxygenase, aryl acylamidase and deaminase enzymes giving 4 aminophenol and hydroquinone as product 22,23,24. Based on the fermentation studies and chemotaxis assay, Bacillus subtilis can be employed for the bioaugmentation of drug polluted area. Conductive polymer/multiwalled carbon nanotube based microbial biosensor using Bacillus subtilis has been reported to have the potential to detect paracetamol even in low concentration26. Considering the reported literature about the acetaminophen using microorganisms, their use is not just limited to degrade the pollutant but also aids in the determination of their concentration.
For the effective treatment of the pharmaceutical industries wastewater, bioaugmentation in a membrane reactor is generally employed where the microorganisms should have the potential to cope up with inhospitable environment27. Report is available on the applicability of the fluidized bed pelleted bioreactor augmented with the fungi Trametes versicolor and Aspergillus luchuensis for the elimination of ketoprofen, metoprolol and ibuprofen28. Bioaugmentation of APAP-degrading organism, Ensifer sp. POKHU to the activated sludge resulted in the reduction of acetaminophen concentration, followed by ecotoxicity level29.
For the biodegradation of acetaminophen in wastewater treatment plants, microorganisms adopted different strategies like internalization of the drugs, cell surface absorption, enzyme assisted mechanism, and biotic- abiotic coupling 30, 31, 32. Due to the rapid and extended eco toxic effects of the pharmaceutical micro pollutants, immediate removal of this pollutants from the source site is needed. Combined approach of Physico and biological processes will ensure the detoxification of wastewater.
Conclusion
Acetaminophen is one of the emerging pharmaceutical micropollutants which must be removed from the water reservoir using an economical and ecofriendly biological approach. Based on the current study, it can be concluded that Bacillus subtilis has the potential to tolerate and degrade high concentrations of acetaminophen. Recovery of acetaminophen degradative product, 4 aminophenol, will be suitable due to its application in dye industries. In the pharmaceutical industrial wastewater treatment plant, emphasis should be given to the degradation of pollutants into the products with industrial applications.
Acknowledgement
Meghmala Waghmode acknowledges Mahajyoti fellowship for the financial assistance.
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding Sources
No any external funding was provided for this research work.
References
- Hu, J., Zhang, L. L., Chen, J. M., and Liu, Y. Degradation of paracetamol by Pseudomonas aeruginosa strain HJ1012. Journal of Environmental Science and Health, Part A.2013; 48(7) 791–799.
CrossRef - Caliman, F. A., and Gavrilescu, M. Pharmaceuticals, personal care products and endocrine disrupting agents in the environment–a review. CLEAN–Soil, Air, Water.2009; 37(4?5) 277-303.
CrossRef - Wu, S., Zhang, L., and Chen, J. Paracetamol in the environment and its degradation by microorganisms. Applied Microbiology and Biotechnology. 2012;96(4), 875–884.
CrossRef - Onesios KM, Yu JT, Bouwer EJ Biodegradation and Removal of Pharmaceuticals and Personal Care Products in Treatment Systems: A Review. Biodegradation 2009;20(4)441–466.
CrossRef - Chandrasekharan, N. V., Dai, H., Roos, K. L. T., Evanson, N. K., Tomsik, J., Elton, T. S., and Simmons, D. L. COX-3, a cyclooxygenase-1 variant inhibited by acetaminophen and other analgesic/antipyretic drugs: Cloning, structure, and expression. Proceedings of the National Academy of Sciences.2002; 99(21)13926–13931.
CrossRef - Zwiener, C. Oxidative treatment of pharmaceuticals in water. Water Research.2000; 34(6) 1881–1885.
CrossRef - Langford, K. H., and Thomas, K. V. Determination of pharmaceutical compounds in hospital effluents and their contribution to wastewater treatment works. Environment International. 200935(5) 766–770.
CrossRef - Akande, M. G. The toxicological evaluation of Sewage Effluents and Pharmaceuticals with the use of Zebrafish as a model organism.2011. https://stud.epsilon.slu.se/3356/1/akande_m_111017.pdf.
- Brillas, E., Sirés, I., Arias, C., Cabot, P. L., Centellas, F., Rodríguez, R. M.,and Garrido, J. A. Mineralization of paracetamol in aqueous medium by anodic oxidation with a boron-doped diamond electrode. Chemosphere. 2005;58(4)399–406.
CrossRef - Waterston, K., Wang, J. W., Bejan, D., and Bunce, N. J. Electrochemical waste water treatment: Electrooxidation of acetaminophen. Journal of Applied Electrochemistry.2006;36(2) 227–232.
CrossRef - Skoumal, M., Cabot, P.-L., Centellas, F., Arias, C., Rodríguez, R. M., Garrido, J. A., & Brillas, E. Mineralization of paracetamol by ozonation catalyzed with Fe2+, Cu2+ and UV light. Applied Catalysis B: Environmental.2006; 66(3–4)228–240.
CrossRef - Yang, L., Yu, L. E., and Ray, M. B. Degradation of paracetamol in aqueous solutions by TiO2 photocatalysis. Water Research.2008;42(13)3480–3488.
CrossRef - Chopra, S., and Kumar, D. Characterization, optimization, and kinetics study of acetaminophen degradation by Bacillus drentensis strain S1 and waste water degradation analysis. Bioresources and Bioprocessing.2020; 7(1)1-18.
CrossRef - Sane, R. T., and Kamat, S. S. Simple Colorimetric Method for Determination of Acetaminophen. Journal of AOAC international.1980; 63(6)1189–1190.
CrossRef - Mulla, S. I., Hu, A., Sun, Q., Li, J., Suanon, F., Ashfaq, M., and Yu, C.P. Biodegradation of sulfamethoxazole in bacteria from three different origins. Journal of Environmental Management.2018; 206(93–102).
CrossRef - Ivshina, I. B., Rychkova, M. I., Vikhareva, E. V., Chekryshkina, L. A., & Mishenina, I. I. Catalysis of the biodegradation of unusable medicines by alkanotrophic rhodococci. Applied Biochemistry and Microbiology.2006;42(4) 392-395.
CrossRef - Zhang, L., Hu, J., Zhu, R., Zhou, Q., and Chen, J. Degradation of paracetamol by pure bacterial cultures and their microbial consortium. Applied microbiology and biotechnology.2013 ;97(8)3687-3698.
CrossRef - Bart DG, Lynn V, Willy V and Nico B Degradation of acetaminophen by Delftia tsuruhatensis and Pseudomonas aeruginosa in a membrane bioreactor. Water Res.2011; 45(1829–1837).
CrossRef - Kumari, T. R., Vidyavathi, M., Asha, S., & Prasad, K. V. S. R. G. Biotransformation of paracetamol by Cunninghamella echinulate. Journal of Pharmaceutical Research.2009; 8(1)1-5.
CrossRef - Chen, R., Liu, X., and Ma, Y. Isolation and identification of acetaminophen degrading strain Shinella sp. HZA2. Journal of Environmental Science and Health, Part B, 2022 (1-6).
CrossRef - Palma, T. L., Magno, G., and Costa, M. C. Biodegradation of paracetamol by some gram-positive bacterial isolates. Current Microbiology.2021;78(7)2774-2786.
CrossRef - dos S Grignet, R., Barros, M. G., Panatta, A. A., Bernal, S. P., Ottoni, J. R., Passarini, M. R., and da CS Gonçalves, C. Medicines as an emergent contaminant: the review of microbial biodegration potential. Folia Microbiologica.2022(1-18).
CrossRef - Lee, S., Park, E. H., Ko, H. J., Bang, W. G., Kim, H. Y., Kim, K. H., and Choi, I. G. Crystal structure analysis of a bacterial aryl acylamidase belonging to the amidase signature enzyme family. Biochemical and biophysical research communications.2015; 467(2)268-274.
CrossRef - ?ur, J., Wojcieszy?ska, D., Hupert-Kocurek, K., Marchlewicz, A., & Guzik, U. Paracetamol–toxicity and microbial utilization. Pseudomonas moorei KB4 as a case study for exploring degradation pathway. Chemosphere.2018;206(192-202).
CrossRef - Rios-Miguel, A. B., Smith, G. J., Cremers, G., van Alen, T., Jetten, M. S., den Camp, H. J. O., & Welte, C. U. (2022). Microbial paracetamol degradation involves a high diversity of novel amidase enzyme candidates. Water Research X.2022; 100152.
CrossRef - Bayram, E., & Akyilmaz, E. (2016). Development of a new microbial biosensor based on conductive polymer/multiwalled carbon nanotube and its application to paracetamol determination. Sensors and Actuators B: Chemical2016; 233(409-418).
CrossRef - Wen, D., Zhang, J., Xiong, R., Liu, R., & Chen, L. (2013). Bioaugmentation with a pyridine-degrading bacterium in a membrane bioreactor treating pharmaceutical wastewater. Journal of Environmental Sciences, 25(11), 2265-2271.
CrossRef - Dalecka, B., Strods, M., Cacivkins, P., Ziverte, E., Rajarao, G. K., & Juhna, T. (2021). Removal of pharmaceutical compounds from municipal wastewater by bioaugmentation with fungi: an emerging strategy using fluidized bed pelleted bioreactor. Environmental Advances, 5, 100086.
CrossRef - Park, S., & Oh, S. (2020). Activated sludge-degrading analgesic drug acetaminophen: Acclimation, microbial community dynamics, degradation characteristics, and bioaugmentation potential. Water research, 182, 115957.
CrossRef - Poddar, K., Sarkar, D., Chakraborty, D., Patil, P. B., Maity, S., & Sarkar, A. (2022). Paracetamol biodegradation by Pseudomonas strain PrS10 isolated from pharmaceutical effluents. International Biodeterioration & Biodegradation, 175, 105490.
CrossRef - Akay, C., & Tezel, U. (2020). Biotransformation of Acetaminophen by intact cells and crude enzymes of bacteria: A comparative study and modelling. Science of the total environment, 703, 134990.
CrossRef - Waghmode, M. S., Patil, N. N., & Bankar, A. V. (2022). Microbial Cell Factories For the Management of Pharmaceutical Micropollutants. International Journal of Ecology and Environmental Sciences, 49(1), 5-16.
CrossRef - Chopra S, Kumar D. Biodegradation, and kinetic analysis of acetaminophen with co-culture of bacterial strains isolated from sewage wastewater. Current microbiology. 2020 Oct;77:3147-57.
CrossRef - https://cake-kinetics.org/







